Why the Public Expects a Coronavirus Vaccine Too Quickly
Executive Summary
- Expectations have been created in the public’s minds regarding the approval of a coronavirus vaccine that is unrealistic.
- We cover how this came about.
Introduction
See our references for this article and related articles at this link.
During the coronavirus, the public had come to expect the quick development of a coronavirus vaccine. It has been common to hear projects of roughly 12 to 18 months.
While performing research for this article, it was shocking to learn of the historical timelines for vaccines’ development. So the question arises, why has the public had its expectations of a vaccine at such a rapid pace?
The Public’s Expectations on a Coronavirus Vaccine
The following is an illustrative quotation on this topic of public expectation.
Almost every day, I hear people making plans around the eventual arrival of a coronavirus vaccine — office reopenings, rescheduled weddings, family reunions and international travel. In recent weeks, colleagues and friends have asked me with growing urgency: “When will we have a vaccine? Will it be any good?” – ProPublica
Journalists Cherry Picking Vaccine Timelines
The desire of government officials to present a rosy scenario regarding the coronavirus is well known. However, there is evidence that the media is selectively choosing to present shorter time estimates to their readers.
This is explained in the following quotation.
More than 30 years ago, I was asked the same question about an AIDS vaccine. Being a realistic optimist, my answer was: I do not expect a vaccine earlier than five years from now. None of the many journalists who asked me that question ever quoted those five years. Far too pessimistic! Other scientists spoke about one or two years. They were cited in the newspapers (and I was no longer harassed by journalists).
Three decades later, we are still waiting for an AIDS vaccine effective enough to be licensed.
I learned a valuable lesson then. Today, when I get asked the same question about the novel coronavirus, I respond by repeating the question with a different emphasis: “When do WE (all) get the coronavirus vaccine?” And there are other important questions to be added: Will it be safe? Will it be reliably protective?”
An average vaccine takes about 10-12 years to be developed. – Jens-Peter Gregersen, DVM
This is reinforced with the following quotation.
The grim truth behind this rosy forecast is that a vaccine probably won’t arrive any time soon. (published in April 2020) Clinical trials almost never succeed.(emphasis added) We’ve never released a coronavirus vaccine for humans before. Our record for developing an entirely new vaccine is at least four years — more time than the public or the economy can tolerate social-distancing orders.
But if there was any time to fast-track a vaccine, it is now. So Times Opinion asked vaccine experts how we could condense the timeline and get a vaccine in the next few months instead of years. – New York Times
Furthermore, such fast vaccine development timelines come with the normal risks associated with time-compressing projects.
There’s a cost to moving so quickly, however. The potential Covid-19 vaccines now in the pipeline might be more likely to fail because of the swift march through the research phase, said Robert van Exan, a cell biologist who has worked in the vaccine industry for decades.
He predicts we won’t see a vaccine approved until at least 2021 or 2022, and even then, “this is very optimistic and of relatively low probability.” – New York Times
This was published in the New York Times in April of 2020 — so it has been published for a while. However, the information contained in this New York Times article received little replay in other media entities.
It almost appears that media entities are incentivized to delude their readers on the coronavirus vaccine timeline.
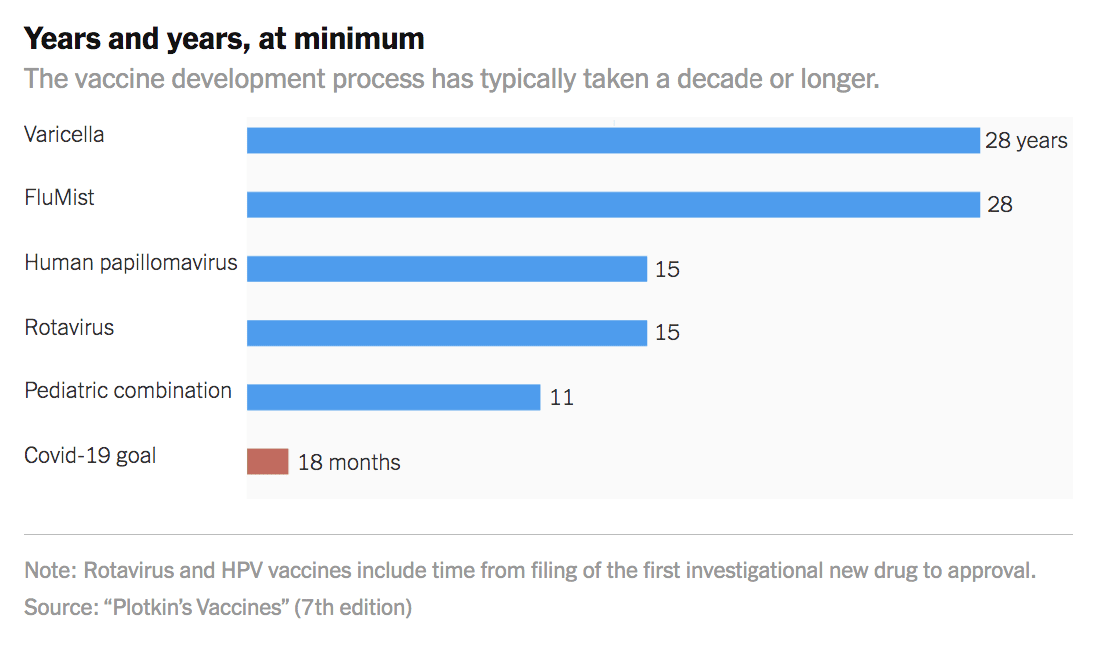
Notice the following graphic, also from the New York Times.
Hmmm…is there a problem with the expectations that are being created for the coronavirus?
How The Fastest Possible Vaccine Development Time Became the Official Estimate
The 12 to 18-month estimate has become the dominant estimate published in media outlets.
If this estimate is so inconsistent with historical vaccine development durations, where did the number come from? It was pointed out by Gregersen, but it is also explained in the following quotation.
Dr. Zervos says the soonest a COVID-19 vaccine could be ready for widespread use will be 18 months from now—and if that’s the case, it will be the fastest a vaccine has ever been developed. – Henry Ford Health Systems
And this quote
Officials like Dr. Anthony S. Fauci, the top infectious disease expert on the Trump administration’s coronavirus task force, estimate a vaccine could arrive in at least 12 to 18 months. – New York Times
So this means Fauci proposed this optimistic timeline. This comment on the New York Times article discusses how little was behind this estimate when he made it.
To Whit, back in March Dr. Fauci made an incredibly casual guess that the soonest a vaccine could be available within a year to eighteen months. From that, a quasi-religious devotion has developed that one WILL be available. Thus, all we’ve got to do is hide away until then, and we’ll all be safe again. – New York Times
This is what I believe happened.
Journalists asked for the fastest time a vaccine could be developed, and this “fastest possible time” became the average time. There is an enormous difference between the average time or and the shortest possible time.
This fact reminds me of some of the interviews I have given to journalists. I have had a similar experience when being interviewed by journalists, in that they will look into my quotes rather than taking my quotes for what they are. That is, they are leading my quotes, looking to pull what they want out of the quotes — as they have a bias in their head, even though they don’t have any domain expertise on the topic. I also have been interviewed by journalists who appear to have a short attention span and can’t process the information I provide to them. I also had a journalist tell me that he normally did not change the story after it was published, even though the name of my company, what it does, and my title were all incorrect.
What Are the Steps for Developing and Bringing Out a New Vaccine?
The steps of developing a vaccine are the following:
#1: Determine the genetic sequence of the virus.
#2: Develop a vaccine using one of a few different strategies.
#3: Start the first clinical trials using healthy, normal volunteers.
#4: Begin the second round of clinical trials in larger populations of people who are at risk for infection.
#5: Approve vaccine for widespread use. – Henry Ford Health Systems
And then, of course, produce and distribute the vaccine.
This quote explains how long some steps take along with their probability of success.